

The UW-Madison ‘One Health Center’ will advance the development and production of vaccines against emerging diseases.

Dr Ella said that Bharat Biotech's nasal vaccine against Covid-19 will soon be available in hospital for inoculation.

NEW DELHI: Bharat Biotech’s Covaxin has proven to be ‘safe, well-tolerated, and highly immunogenic’ in children in Phase II/III studies, the results of which are published in the prestigious Lancet journal. The study said that the data from over 50 million doses given to children in India reveals that side effects are minimal. The trials […]
NEW DELHI: Bharat Biotech International Limited confirms that COVAXIN successfully conducted the stage II/III study on children. The trials have proven to be safe, well tolerated and immunogenic among children. COVAXIN trails show even better response in children aged between 2-18 years than adults. Bharat Biotech conducted the phase II/III multicenter studies to evaluate the […]
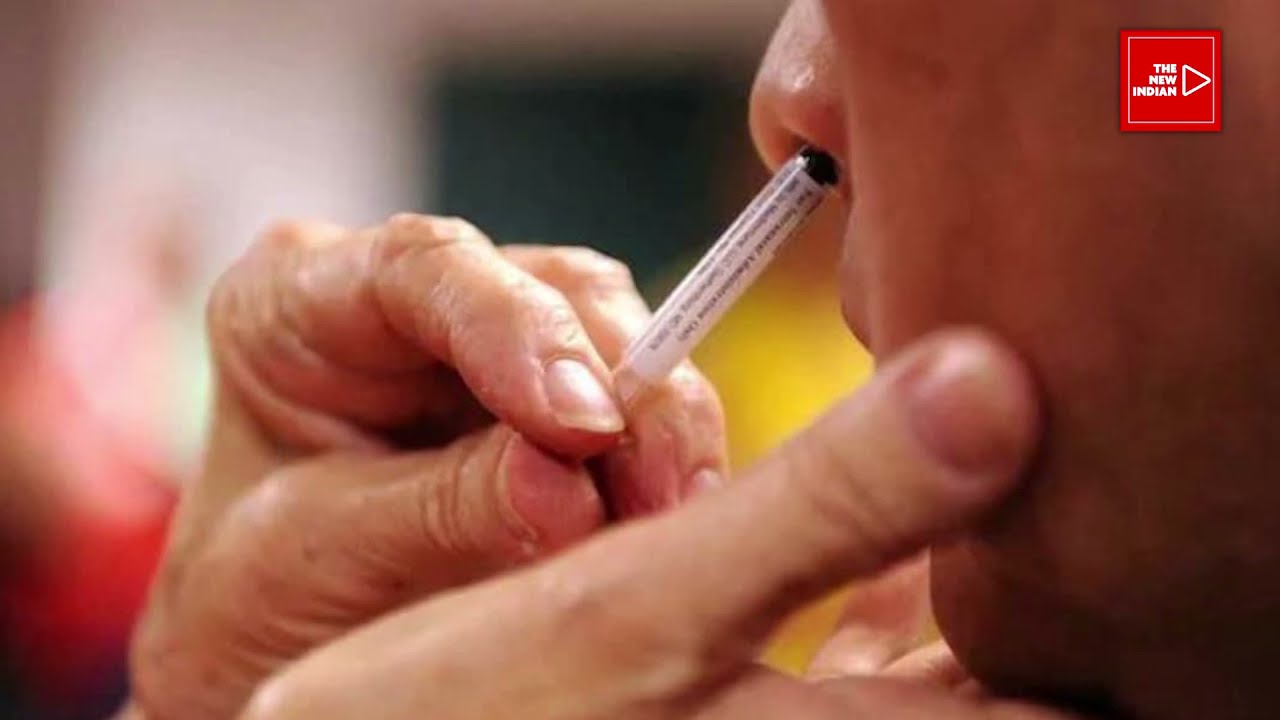
Even as India celebrated the 100 Crore vaccine dose mark last week, Dr. Krishna Ella who is the founder of Bharat Biotech, maker of indegenious ‘Covaxin’ told The New Indian that his company will be bringing the first nasal Covid vaccine and is expected to launch within the first few months of 2022. Dr. Ella […]

NEW DELHI: In the part two of an extensive interview with The New Indian’s Executive Editor, Dr Krishna Ella of Bharat Biotech speaks on the vaccine efficacy and shares his plans of ramping up the production to produce 1 to 1.5 cr more vaccine doses to meet the annual target of 100 cr. EXCLUSIVE: PART […]

NEW DELHI: In the very first interaction hours ahead of WHO permission to issue international certification, Dr Krishna Ella of Bharat Biotech shares with The New Indian his plans for the launch of nasal vaccine, his disappointment with false news over Covaxin and global politics of over homegrown vaccine. Rohan Dua: Despite creating an indigenous […]